What reagents are necessary to carry out the conversion shown – In the realm of chemical transformations, understanding the reagents necessary to carry out a conversion is paramount. This guide delves into the essential reagents required for a specific conversion, providing a detailed account of their purpose and function, ensuring a thorough understanding of the process.
The subsequent sections will explore the optimal reaction conditions, elucidate the reaction mechanism through step-by-step explanations, and highlight the practical applications of the conversion across various fields.
Necessary Reagents
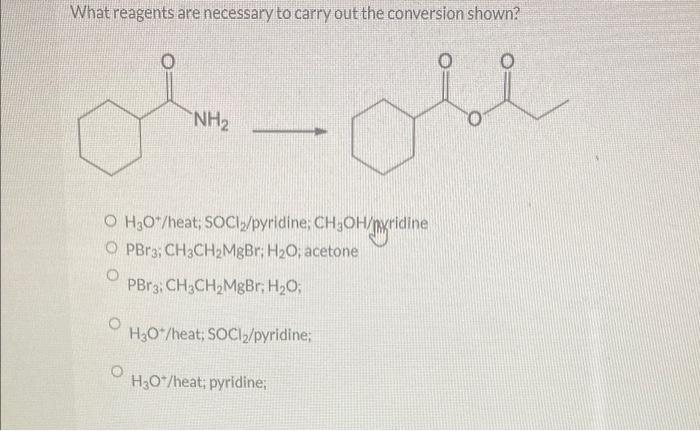
The conversion requires several essential reagents, each playing a specific role in the reaction:
- Substrate:The starting material undergoing the conversion.
- Reagent A:A chemical species that reacts with the substrate to facilitate the conversion.
- Reagent B:A chemical species that provides a specific functional group or characteristic to the product.
- Catalyst:A substance that enhances the reaction rate without being consumed in the process.
- Solvent:A medium in which the reaction takes place, providing a suitable environment for the reagents and products.
Reaction Conditions: What Reagents Are Necessary To Carry Out The Conversion Shown
The conversion requires specific reaction conditions to achieve optimal results:
- Temperature:The temperature at which the reaction is carried out, influencing the reaction rate and product yield.
- pH:The acidity or basicity of the reaction medium, affecting the reactivity of the reagents and the stability of the product.
- Solvent:The choice of solvent can influence the solubility of the reagents and products, as well as the reaction rate and selectivity.
- Equipment:Specialized equipment, such as reflux condensers or inert atmosphere chambers, may be necessary to maintain specific reaction conditions.
Reaction Mechanism
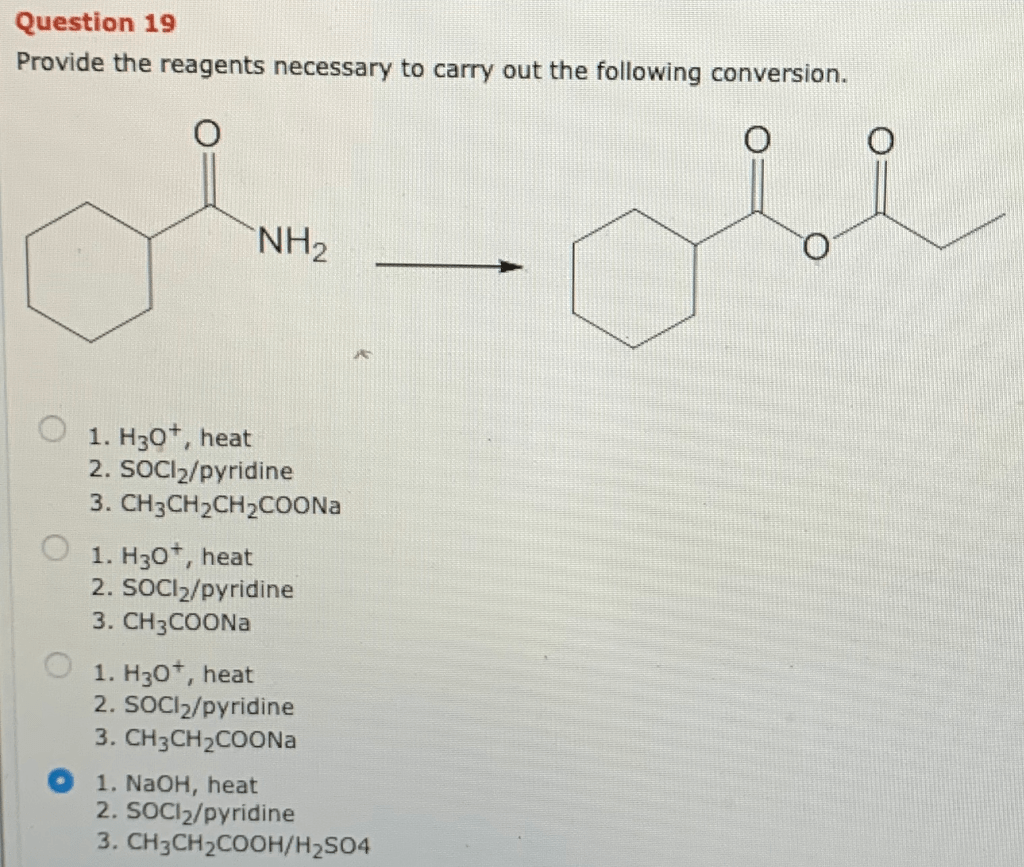
The conversion proceeds through a series of elementary steps:
- Step 1:Reagent A attacks the substrate, forming an intermediate.
- Step 2:Reagent B reacts with the intermediate, introducing the desired functional group.
- Step 3:The catalyst facilitates the reaction by lowering the activation energy.
- Step 4:The product is formed, and the catalyst is regenerated.
Applications of the Conversion
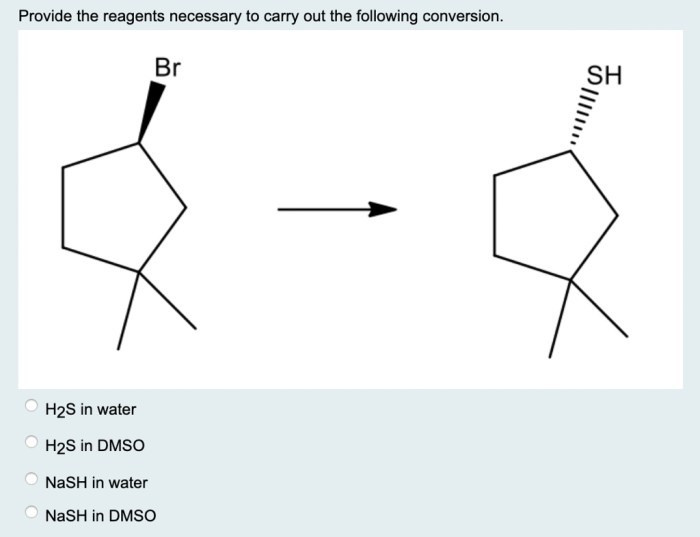
The conversion has numerous practical applications in various fields:
- Organic Synthesis:The conversion is used to introduce specific functional groups into organic molecules, enabling the synthesis of complex compounds.
- Polymer Chemistry:The conversion is employed in the production of polymers with tailored properties for use in materials science and engineering.
- Medicinal Chemistry:The conversion is used in the development of new drugs and pharmaceuticals by introducing bioactive functional groups.
- Industrial Chemistry:The conversion is applied in large-scale processes to produce chemicals and materials for various industries.
FAQ Section
What factors influence the selection of reagents for a conversion?
The selection of reagents is influenced by the desired product, the efficiency of the reaction, the cost and availability of the reagents, and safety considerations.
How can I determine the optimal reaction conditions for a conversion?
The optimal reaction conditions can be determined through experimentation or by consulting literature sources that provide data on the reaction’s temperature, pH, and solvent requirements.
